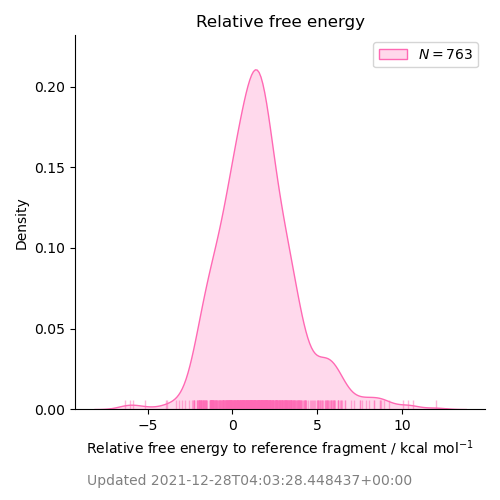
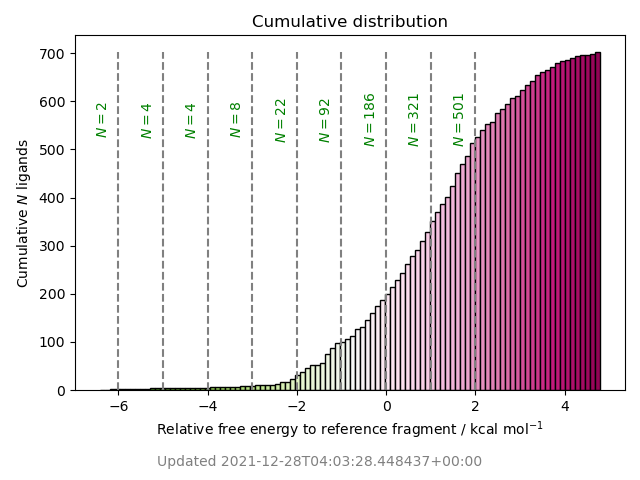
COVID Moonshot Sprint 10 for P1 pocket replacement based on x10959 (ADA-UCB-6c2cb422-1) to optimize substituents in the P1 pocket with Mpro dimer and neutral Cys145:His41 catalytic dyad
| Rank | Compound | SMILES | ΔG / kcal mol-1 | pIC50 | |
|---|---|---|---|---|---|
| 1 | DAR-DIA-56cf811e-1 |
|
C#CC(=O)N(c1cncc2c1cccc2)C(=O)Cc3cccc(c3)Cl | −13.9 ± 0.1 | 10.1 ± 0.1 |
| 2 | TRY-UNI-9f475305-8 |
|
Cc1cc(no1)CN(C(=O)C(C)c2cccc(c2)Cl)C(=O)NC3CC3 | −13.5 ± 0.2 | 9.9 ± 0.1 |
| 3 | PET-UNK-12d8d43f-2 |
|
COC(=O)N(c1cncc2c1cccc2)C(=O)Cc3cccc(c3)Cl | −12.7 ± 0.2 | 9.2 ± 0.1 |
| 4 | JOH-UNI-a38a7bdd-7 |
|
Cc1ccncc1N(C(=O)Cc2cccc(c2)Cl)C(=O)C3CC3C(F)(F)F | −10.8 ± 0.1 | 7.9 ± 0.1 |
| 5 | EDJ-MED-c8e7a002-17 |
|
c1cc(cc(c1)Cl)CC(=O)Nc2c3nnc(n3cnc2Cl)O | −10.5 ± 0.1 | 7.66 ± 0.07 |
| 6 | BAR-COM-0f94fc3d-24 |
|
c1ccc2c(c1)ncn2NC(=O)C(c3cccc(c3)Cl)(F)F | −10.4 ± 0.1 | 7.55 ± 0.08 |
| 7 | EDJ-MED-ee07cf00-13 |
|
Cc1nc2n(n1)C[C@@H](CC2)C(=O)NC(c3cccc(c3)Cl)C(=O)Nc4cncc5c4cccc5 | −10.3 ± 0.2 | 7.5 ± 0.1 |
| 8 | EDJ-MED-c8e7a002-8 |
|
c1cc(cc(c1)Cl)CC(=O)Nc2c3nnnn3cnc2Cl | −9.8 ± 0.1 | 7.17 ± 0.10 |
| 9 | RUB-POS-1325a9ea-18 |
|
c1ccc2c(c1)cnc(c2NC(=O)Cc3cccc(c3)Cl)F | −9.82 ± 0.03 | 7.16 ± 0.02 |
| 10 | PET-UNK-1320d94d-9 |
|
c1ccc2c(c1)cncc2NC(=O)[C@@H](c3cccc(c3)Cl)NC(=O)[C@@H]4CCC[C@@H](O4)C(=O)N5CCCC5 | −9.7 ± 0.2 | 7.0 ± 0.2 |
| 11 | EDJ-MED-c8e7a002-1 |
|
COc1ccc2c(c1)onc2NC(=O)Cc3cccc(c3)Cl | −9.6 ± 0.1 | 7.02 ± 0.09 |
| 12 | PET-UNK-ac320b15-7 |
|
c1ccc2c(c1)cncc2NC(=O)[C@@H](c3cccc(c3)Cl)NC(=O)[C@@H]4CC[C@H](O4)C(=O)N5CCCC5 | −9.6 ± 0.2 | 7.0 ± 0.1 |
| 13 | EDJ-MED-076d6e64-1 |
|
c1cc(cc(c1)Cl)CC(=O)Nc2cncn3c2nnc3 | −9.6 ± 0.1 | 6.96 ± 0.08 |
| 14 | BAR-COM-ebf5acce-14 |
|
Cc1ccncc1N(CCn2cccn2)C(=O)Cc3cccc(c3)Cl | −9.5 ± 0.2 | 7.0 ± 0.1 |
| 15 | JAN-GHE-5a013bed-9 |
|
c1cc(cc(c1)Cl)CC(=O)Nc2cncnc2 | −9.5 ± 0.1 | 6.93 ± 0.09 |
| 16 | PET-UNK-1320d94d-21 |
|
CN(C)C(=O)[C@H]1CC[C@H](O1)C(=O)N(C)[C@H](c2cccc(c2)Cl)C(=O)Nc3cncc4c3cccc4 | −9.5 ± 0.2 | 6.9 ± 0.2 |
| 17 | JAN-GHE-83b26c96-7 |
|
Cc1c(cncn1)NC(=O)C(c2cccc(c2)Cl)C(C)C | −9.4 ± 0.2 | 6.9 ± 0.1 |
| 18 | MIC-UNK-c66144cb-3 |
|
c1ccc2c(c1)cncc2NC(=O)C(CCC3CCCCC3)c4cccc(c4)Cl | −9.4 ± 0.2 | 6.9 ± 0.2 |
| 19 | VLA-UNK-dc1d9354-1 |
|
c1ccc2c(c1)[nH]c(=O)n2NC(=O)Cc3cccc(c3)Cl | −9.4 ± 0.1 | 6.8 ± 0.1 |
| 20 | PET-UNK-1320d94d-29 |
|
CN([C@H](c1cccc(c1)Cl)C(=O)Nc2cncc3c2cccc3)C(=O)[C@@H]4CCC[C@@H](O4)C(=O)N5CCCC5 | −9.2 ± 0.2 | 6.7 ± 0.2 |
| 21 | JAN-GHE-5a013bed-4 |
|
c1cc(cc(c1)Cl)CC(=O)Nn2cn[nH]c2=O | −9.2 ± 0.1 | 6.70 ± 0.09 |
| 22 | ALP-POS-3b848b35-4 |
|
c1ccc2c(c1)cncc2NC(=O)C(CCC3CCCCC3)c4cccc(c4)Cl | −9.2 ± 0.2 | 6.7 ± 0.1 |
| 23 | VLA-UNK-411a133b-1 |
|
Cn1nc(nn1)NC(=O)Cc2cccc(c2)Cl | −9.2 ± 0.1 | 6.68 ± 0.08 |
| 24 | JAN-GHE-f4ca5a00-20 |
|
CCCCC(c1cccc(c1)Cl)C(=O)Nn2c(cnn2)C3CC3 | −9.1 ± 0.2 | 6.6 ± 0.1 |
| 25 | PET-UNK-1320d94d-1 |
|
CN(C)C(=O)[C@H]1CC[C@H](O1)C(=O)N[C@H](c2cccc(c2)Cl)C(=O)Nc3cncc4c3cccc4 | −9.1 ± 0.2 | 6.6 ± 0.1 |
| 26 | MAT-POS-bfb445d4-2 |
|
c1ccc2c(c1)nnn2NC(=O)Cc3cccc(c3)Cl | −9.11 ± 0.07 | 6.63 ± 0.05 |
| 27 | MIC-UNK-5a93dd5f-12 |
|
c1ccc2c(c1)cncc2NC(=O)C(c3cccc(c3)Cl)N4CCC(C4)N5CCCCC5 | −9.1 ± 0.1 | 6.62 ± 0.08 |
| 28 | MIC-UNK-d36ab305-4 |
|
CN(C)c1ccc(cc1)C(c2cccc(c2)Cl)C(=O)Nc3cncc4c3cccc4 | −9.1 ± 0.2 | 6.6 ± 0.1 |
| 29 | DAR-DIA-076fb6ea-6 |
|
Cc1ccncc1N(C#CC#N)C(=O)Cc2cccc(c2)Cl | −9.0 ± 0.2 | 6.6 ± 0.1 |
| 30 | JAN-GHE-f4ca5a00-18 |
|
CCCCC(c1cccc(c1)Cl)C(=O)Nn2cncc2C3CC3 | −9.0 ± 0.2 | 6.6 ± 0.1 |
| 31 | MIC-UNK-c66144cb-1 |
|
c1ccc2c(c1)cncc2NC(=O)C(CCc3cccc(c3)F)c4cccc(c4)Cl | −8.9 ± 0.2 | 6.5 ± 0.2 |
| 32 | PET-UNK-ac320b15-5 |
|
c1ccc2c(c1)cncc2NC(=O)[C@@H](c3cccc(c3)Cl)NC(=O)[C@H]4CC[C@@H](O4)C(=O)N5CCCC5 | −8.9 ± 0.2 | 6.5 ± 0.2 |
| 33 | EDJ-MED-c8e7a002-13 |
|
c1cc(cc(c1)Cl)CC(=O)Nc2c3c(cccc3F)n[nH]2 | −8.9 ± 0.1 | 6.46 ± 0.10 |
| 34 | PET-UNK-1320d94d-25 |
|
CN([C@H](c1cccc(c1)Cl)C(=O)Nc2cncc3c2cccc3)C(=O)[C@@H]4CC[C@@H](O4)C(=O)N5CCC5 | −8.9 ± 0.2 | 6.5 ± 0.1 |
| 35 | JOH-UNI-a38a7bdd-3 |
|
Cc1ccncc1N(C(=O)Cc2cccc(c2)Cl)C(=O)C(F)(F)F | −8.8 ± 0.2 | 6.4 ± 0.1 |
| 36 | PET-UNK-1320d94d-5 |
|
c1ccc2c(c1)cncc2NC(=O)[C@@H](c3cccc(c3)Cl)NC(=O)[C@@H]4CC[C@@H](O4)C(=O)N5CCC5 | −8.8 ± 0.2 | 6.4 ± 0.1 |
| 37 | PET-UNK-6314f867-3 |
|
c1cc(cc(c1)Cl)CC(=O)Nc2cncc3c2c(ccc3)C#N | −8.8 ± 0.1 | 6.43 ± 0.09 |
| 38 | TRY-UNI-9f475305-3 |
|
Cc1ccncc1NC(=O)C(c2cccc(c2)Cl)c3nnc(s3)C | −8.8 ± 0.1 | 6.42 ± 0.08 |
| 39 | MAT-POS-bb423b95-8 |
|
c1cc(cc(c1)Cl)CC(=O)Nc2cncc3c2ccc(c3)O | −8.8 ± 0.1 | 6.42 ± 0.09 |
| 40 | MIC-UNK-c66144cb-2 |
|
c1ccc2c(c1)cncc2NC(=O)C(CCc3ccc(cc3)F)c4cccc(c4)Cl | −8.8 ± 0.2 | 6.4 ± 0.1 |
| 41 | SAM-UNK-2684b532-3 |
|
Cc1ccncc1NC(=O)C(c2cccc(c2)Cl)c3nnc(s3)C | −8.8 ± 0.1 | 6.40 ± 0.08 |
| 42 | PET-UNK-1320d94d-3 |
|
c1ccc2c(c1)cncc2NC(=O)[C@@H](c3cccc(c3)Cl)NC(=O)[C@@H]4CC[C@@H](O4)C(=O)N5CCOCC5 | −8.8 ± 0.2 | 6.4 ± 0.2 |
| 43 | EDJ-MED-97c1bf5c-2 |
|
COc1c(c2nnnn2cn1)NC(=O)Cc3cccc(c3)Cl | −8.8 ± 0.1 | 6.39 ± 0.10 |
| 44 | PET-UNK-5ecb6237-1 |
|
c1ccc2c(c1)cncc2N(C#N)C(=O)Cc3cccc(c3)Cl | −8.8 ± 0.2 | 6.4 ± 0.1 |
| 45 | DAR-DIA-076fb6ea-3 |
|
COC(=O)/C=C/C(=O)N(c1cncc2c1cccc2)C(=O)Cc3cccc(c3)Cl | −8.7 ± 0.2 | 6.3 ± 0.2 |
| 46 | JAN-GHE-83b26c96-15 |
|
Cc1ccncc1N(C)C(=O)Cc2cccc(c2)Cl | −8.7 ± 0.2 | 6.3 ± 0.1 |
| 47 | DAR-DIA-076fb6ea-7 |
|
Cc1ccncc1N(C(=O)Cc2cccc(c2)Cl)C(=O)C=C=C | −8.7 ± 0.2 | 6.3 ± 0.1 |
| 48 | EDJ-MED-ee07cf00-11 |
|
c1ccc2c(c1)cncc2NC(=O)C(c3cccc(c3)Cl)NC(=O)C4CCC(O4)C(=O)N5CCCC5 | −8.6 ± 0.1 | 6.30 ± 0.08 |
| 49 | PET-UNK-6314f867-2 |
|
c1cc(cc(c1)Cl)CC(=O)Nc2cncc3c2ccc(c3)C#N | −8.6 ± 0.2 | 6.2 ± 0.1 |
| 50 | JIN-POS-6dc588a4-16 |
|
c1cc(cc(c1)Cl)CC(=O)Nc2c3ccncc3on2 | −8.5 ± 0.1 | 6.23 ± 0.08 |
| 51 | PET-UNK-b38839dc-2 |
|
COc1cc2c(cc1Cl)cncc2NC(=O)Cc3cccc(c3)Cl | −8.5 ± 0.1 | 6.2 ± 0.1 |
| 52 | PET-UNK-ac320b15-9 |
|
c1ccc2c(c1)cncc2NC(=O)[C@@H](c3cccc(c3)Cl)NC(=O)[C@H]4CC[C@H](O4)C(=O)N5CCCC5 | −8.5 ± 0.2 | 6.2 ± 0.1 |
| 53 | VLA-UNK-9a7dc93f-7 |
|
COC(c1cccc(c1)Cl)C(=O)Nc2cncc3c2cccc3 | −8.51 ± 0.05 | 6.20 ± 0.03 |
| 54 | ROB-UNI-daaf9793-3 |
|
c1ccc2c(c1)CC(=CC2=O)NC(=O)Cc3cccc(c3)Cl | −8.5 ± 0.1 | 6.2 ± 0.1 |
| 55 | JIN-POS-6dc588a4-17 |
|
c1cc(cc(c1)Cl)CC(=O)Nc2cc3nccn3cn2 | −8.5 ± 0.2 | 6.2 ± 0.1 |
| 56 | PET-UNK-1320d94d-7 |
|
c1ccc2c(c1)cncc2NC(=O)[C@@H](c3cccc(c3)Cl)NC(=O)[C@@H]4C[C@@H](O4)C(=O)N5CCCC5 | −8.5 ± 0.2 | 6.2 ± 0.2 |
| 57 | JAN-GHE-5a013bed-2 |
|
c1ccc2c(c1)ncn2NC(=O)Cc3cccc(c3)Cl | −8.41 ± 0.07 | 6.13 ± 0.05 |
| 58 | EDJ-MED-ee07cf00-2 |
|
Cc1c(c(=O)n2c(n1)cc[nH]2)CC(=O)NC(c3cccc(c3)Cl)C(=O)Nc4cncc5c4cccc5 | −8.4 ± 0.2 | 6.1 ± 0.1 |
| 59 | MAT-POS-a13804f0-4 |
|
c1ccc2c(c1)cncc2NC(=O)C(c3cccc(c3)Cl)NC(=O)[C@@H]4CC[C@@H](O4)C(=O)N5CCCC5 | −8.35 ± 0.07 | 6.09 ± 0.05 |
| 60 | JAN-GHE-f4ca5a00-17 |
|
c1cc(cc(c1)Cl)CC(=O)Nn2c(cnn2)C3CC3 | −8.4 ± 0.1 | 6.09 ± 0.08 |
| 61 | MIC-UNK-5a93dd5f-3 |
|
c1ccc2c(c1)cncc2NC(=O)C(c3cccc(c3)Cl)N4CCC5CCCCC5C4 | −8.35 ± 0.08 | 6.08 ± 0.06 |
| 62 | PET-UNK-10339a1d-1 |
|
CN(C(c1cccc(c1)Cl)C(=O)Nc2cncc3c2cccc3)C(=O)[C@@H]4CC[C@@H](O4)C(=O)N5CCCC5 | −8.3 ± 0.2 | 6.1 ± 0.1 |
| 63 | JOH-UNI-ee5ed7c8-6 |
|
c1ccc2c(c1)cncc2NC(=O)C(CC#N)c3cccc(c3)Cl | −8.3 ± 0.1 | 6.07 ± 0.07 |
| 64 | PET-UNK-1320d94d-30 |
|
CN([C@@H](c1cccc(c1)Cl)C(=O)Nc2cncc3c2cccc3)C(=O)[C@@H]4CCC[C@@H](O4)C(=O)N5CCCC5 | −8.3 ± 0.2 | 6.1 ± 0.1 |
| 65 | MIC-UNK-5a93dd5f-2 |
|
c1ccc2c(c1)cncc2NC(=O)C(c3cccc(c3)Cl)N4CC5CCCCC5C4 | −8.3 ± 0.2 | 6.1 ± 0.1 |
| 66 | MIC-UNK-d36ab305-1 |
|
CC(=O)Nc1ccc(cc1)C(c2cccc(c2)Cl)C(=O)Nc3cncc4c3cccc4 | −8.3 ± 0.1 | 6.06 ± 0.10 |
| 67 | EDJ-MED-c8e7a002-7 |
|
c1cc(cc(c1)Cl)CC(=O)Nc2c3nncn3cnc2Cl | −8.3 ± 0.1 | 6.0 ± 0.1 |
| 68 | MAT-POS-f7918075-3 |
|
c1cc(cc(c1)Cl)CC(=O)Nc2cncc3c2cccn3 | −8.29 ± 0.04 | 6.04 ± 0.03 |
| 69 | PET-UNK-1320d94d-10 |
|
c1ccc2c(c1)cncc2NC(=O)[C@H](c3cccc(c3)Cl)NC(=O)[C@@H]4CCC[C@@H](O4)C(=O)N5CCCC5 | −8.2 ± 0.2 | 6.0 ± 0.2 |
| 70 | MIC-UNK-50cce87d-3 |
|
Cc1cccc2c1c(cnc2)NC(=O)Cc3cccc(c3)Cl | −8.24 ± 0.07 | 6.00 ± 0.05 |
| 71 | ADA-UCB-6c2cb422-11 |
|
COc1ccccc1CO[C@@H](c2cccc(c2)Cl)C(=O)Nc3cc(=O)[nH]c4c3cccc4 | −8.2 ± 0.2 | 6.0 ± 0.2 |
| 72 | JOH-UNI-3fc3434e-7 |
|
CN(c1cncc2c1cccc2)C(=O)C(CC#N)c3cccc(c3)Cl | −8.2 ± 0.2 | 6.0 ± 0.2 |
| 73 | PET-UNK-6c2be958-1 |
|
c1ccc2c(c1)cncc2N(C(=O)Cc3cccc(c3)Cl)C(=O)CCl | −8.2 ± 0.2 | 6.0 ± 0.1 |
| 74 | MAK-UNK-c749d764-22 |
|
CC(CNCC1CCCO1)C(c2cccc(c2)Cl)C(=O)Nc3cncc4c3cccc4 | −8.2 ± 0.2 | 5.9 ± 0.1 |
| 75 | PET-UNK-e274cad4-1 |
|
Cn1c2c(cncc2NC(=O)Cc3cccc(c3)Cl)cn1 | −8.2 ± 0.1 | 5.95 ± 0.09 |
| 76 | PET-UNK-1320d94d-22 |
|
CN(C)C(=O)[C@H]1CC[C@H](O1)C(=O)N(C)[C@@H](c2cccc(c2)Cl)C(=O)Nc3cncc4c3cccc4 | −8.1 ± 0.2 | 5.9 ± 0.1 |
| 77 | JOH-UNI-a38a7bdd-1 |
|
c1ccc2c(c1)cncc2N(C(=O)Cc3cccc(c3)Cl)C(=O)C(F)(F)F | −8.1 ± 0.2 | 5.9 ± 0.1 |
| 78 | MAT-POS-f7918075-7 |
|
c1cc(cc(c1)Cl)CC(=O)Nc2cncc3c2CC(=O)N3 | −8.1 ± 0.1 | 5.9 ± 0.1 |
| 79 | EDJ-MED-00c1612e-1 |
|
COc1ccc2cncc(c2c1)NC(=O)Cc3cccc(c3)Cl | −8.12 ± 0.08 | 5.91 ± 0.05 |
| 80 | MAK-UNK-ffc90da7-4 |
|
CC(CNCC1CCCO1)C(c2cccc(c2)Cl)C(=O)Nc3cncc4c3cccc4 | −8.1 ± 0.2 | 5.9 ± 0.1 |
| 81 | PET-UNK-ac320b15-6 |
|
c1ccc2c(c1)cncc2NC(=O)[C@H](c3cccc(c3)Cl)NC(=O)[C@H]4CC[C@@H](O4)C(=O)N5CCCC5 | −8.1 ± 0.3 | 5.9 ± 0.2 |
| 82 | MAT-POS-29385cc1-4 |
|
Cc1cc(cc2c1c(cnc2)NC(=O)Cc3cccc(c3)Cl)F | −8.10 ± 0.10 | 5.90 ± 0.07 |
| 83 | PET-UNK-8df914d1-1 |
|
COc1ccc2cncc(c2c1)NC(=O)Cc3cccc(c3)Cl | −8.1 ± 0.1 | 5.87 ± 0.09 |
| 84 | MAK-UNK-c749d764-6 |
|
CC(CNCCCO)C(c1cccc(c1)Cl)C(=O)Nc2cncc3c2cccc3 | −8.0 ± 0.2 | 5.9 ± 0.2 |
| 85 | PET-UNK-12d8d43f-1 |
|
c1ccc2c(c1)cncc2N(C=O)C(=O)Cc3cccc(c3)Cl | −8.0 ± 0.1 | 5.85 ± 0.10 |
| 86 | MAK-UNK-ffc90da7-1 |
|
CC(CNCCCO)C(c1cccc(c1)Cl)C(=O)Nc2cncc3c2cccc3 | −8.0 ± 0.2 | 5.8 ± 0.2 |
| 87 | JIN-POS-6dc588a4-11 |
|
c1cc(cc(c1)Cl)CC(=O)Nc2cncc3c2ccc(c3)F | −7.99 ± 0.07 | 5.82 ± 0.05 |
| 88 | PET-UNK-1320d94d-27 |
|
CN([C@H](c1cccc(c1)Cl)C(=O)Nc2cncc3c2cccc3)C(=O)[C@@H]4C[C@@H](O4)C(=O)N5CCCC5 | −8.0 ± 0.2 | 5.8 ± 0.2 |
| 89 | PET-UNK-1320d94d-2 |
|
CN(C)C(=O)[C@H]1CC[C@H](O1)C(=O)N[C@@H](c2cccc(c2)Cl)C(=O)Nc3cncc4c3cccc4 | −8.0 ± 0.2 | 5.8 ± 0.1 |
| 90 | VLA-UCB-05e51b3f-15 |
|
c1ccc2c(c1)cncc2N(C(=O)Cc3cccc(c3)Cl)C(=O)CCl | −7.9 ± 0.2 | 5.8 ± 0.1 |
| 91 | MIC-UNK-50cce87d-2 |
|
c1cc(cc(c1)Cl)CC(=O)Nc2cncc3c2c(ccc3)Cl | −7.9 ± 0.1 | 5.76 ± 0.08 |
| 92 | RUB-POS-1325a9ea-4 |
|
Cc1ccc2cncc(c2c1)NC(=O)Cc3cccc(c3)Cl | −7.91 ± 0.07 | 5.76 ± 0.05 |
| 93 | PET-UNK-1320d94d-26 |
|
CN([C@@H](c1cccc(c1)Cl)C(=O)Nc2cncc3c2cccc3)C(=O)[C@@H]4CC[C@@H](O4)C(=O)N5CCC5 | −7.9 ± 0.2 | 5.8 ± 0.1 |
| 94 | EDJ-MED-ee07cf00-7 |
|
Cc1c(c(=O)ccn1CC(=O)NC(c2cccc(c2)Cl)C(=O)Nc3cncc4c3cccc4)O | −7.9 ± 0.3 | 5.7 ± 0.2 |
| 95 | PET-UNK-a692de38-1 |
|
c1ccc2c(c1)cncc2N(C(=O)Cc3cccc(c3)Cl)NC#N | −7.9 ± 0.2 | 5.7 ± 0.2 |
| 96 | MIC-UNK-5a93dd5f-1 |
|
c1ccc2c(c1)cncc2NC(=O)C(c3cccc(c3)Cl)N4CC5CCCC5C4 | −7.9 ± 0.1 | 5.7 ± 0.1 |
| 97 | EDJ-MED-cf4b0d25-3 |
|
c1ccc2c(c1)cncc2NC(=O)C(c3cccc(c3)Cl)NC(=O)[C@@H]4CCCO4 | −7.84 ± 0.06 | 5.71 ± 0.05 |
| 98 | RUB-POS-1325a9ea-15 |
|
Cn1c2cncc(c2cn1)NC(=O)Cc3cccc(c3)Cl | −7.8 ± 0.1 | 5.71 ± 0.10 |
| 99 | EDG-MED-0da5ad92-18 |
|
Cc1ccnc(c1NC(=O)Cc2cccc(c2)Cl)N | −7.81 ± 0.08 | 5.69 ± 0.06 |
| 100 | MAT-POS-29385cc1-2 |
|
COc1cc2cncc(c2cc1F)NC(=O)Cc3cccc(c3)Cl | −7.8 ± 0.1 | 5.69 ± 0.09 |